When you picture a robot, your brain automatically jumps to metallic joints, whirring gears, silicone chips, and heavy battery packs. Now, throw all of that out the window. What if a robot didn’t look like a shiny mechanical rover, but instead looked like a tiny, fleshy blob of biological jelly that can swim, push payloads, and heal its own wounds? It sounds like a premise ripped straight from a bizarre sci-fi thriller, but it’s happening right now in active laboratories.

In 2020, a team of biologists and computer scientists from Tufts University and the University of Vermont completely blurred the line between organism and machine. By scraping skin and heart stem cells from an African clawed frog embryo, they built something entirely new. It is not a traditional robot, nor is it a naturally occurring species. It is a completely programmable, biological machine designed by an artificial intelligence.
Designing Xenobots Inside a Supercomputer
Before the scientists even picked up a petri dish, they used an evolutionary algorithm on a massive supercomputer to run thousands of random designs for these new biological machines. The AI simulated how different configurations of skin and heart cells would behave in a watery environment. The basic building blocks were simple: skin cells act as a rigid structural frame, while heart muscle cells—which naturally expand and contract on their own—act as microscopic motors.
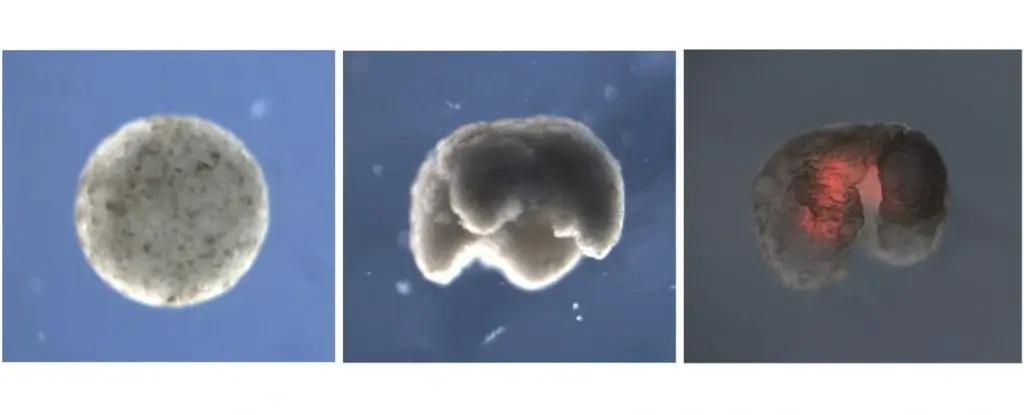
The computer mercilessly tested these 3D designs, keeping the ones that successfully moved forward and tossing the failures. Eventually, it spit out a final blueprint that looked like a tiny, four-legged blob. Only then did the human biologists take over. Using microscopic tweezers and tiny electrodes, they carefully shaped actual frog cells to perfectly match the AI’s design, successfully birthing the first generation of living robots made from frog cells.
The Mechanical Biology of Swimming Blobs
Unlike a remote-controlled drone, these biological machines do not need an external power source, a motherboard, or a complex control chip. They are completely powered by the natural energy stored within the embryonic cells. This biological battery holds enough fuel for them to survive and operate independently for about a week to ten days in a freshwater dish.
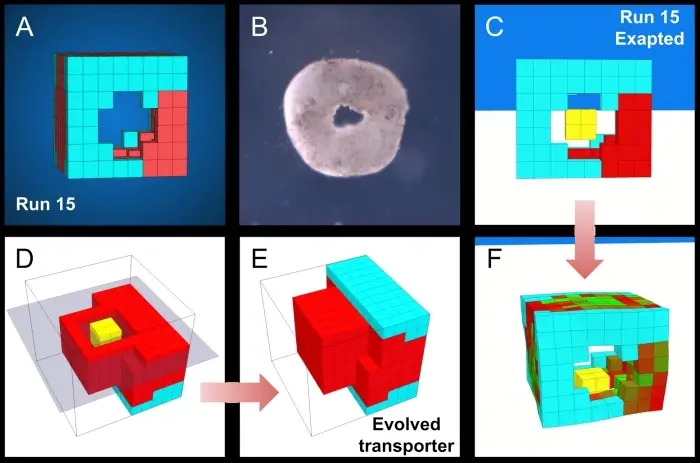
Because their “engines” are made of pulsating heart muscle tissue, they naturally beat and contract. By shaping the blob in highly specific ways, the scientists channeled those random contractions into coordinated, forward movement. They can swim in circles, push microscopic objects (like medicine payloads) into designated piles, and even work together in synchronized swarms. The scientists essentially harnessed the raw mechanical power of biology to do heavy lifting at a microscopic level.
A Machine That Fixes Itself
Perhaps the most amazing advantage of these living robots made from frog cells is their sheer resilience. If you drop your smartphone or crack a mechanical drone, it is broken until a human manually replaces the hardware. Biology, however, has spent billions of years perfecting the art of survival and cellular repair.
When the researchers sliced one of these tiny biological machines almost entirely in half to see what would happen, the blob didn’t die or permanently break down. Instead, the cells immediately began to pull themselves back together, zipping the massive wound closed and going right back to their normal swimming routine. Try getting your household robot vacuum to do that after you hit it with a hammer.
Conclusion
Right now, these blobs are less than a millimeter wide and relatively simple in their functions. But the implications for the future are absolutely wild. Imagine deploying custom-built biological machines into the ocean to hunt down and digest microplastics, or injecting them into human arteries to safely scrape away dangerous plaque and deliver targeted cancer drugs directly to a tumor. Once their mission is over, they simply degrade back into harmless dead skin cells, leaving zero pollution behind. They force us to completely rethink the boundary between biology and technology, proving that life itself might be the ultimate engineering material.
References:
Wired — Meet the Xenobot: The World’s First Living, Self-Healing Machine
Scientific American — World’s First Living Robots Can Now Reproduce
National Geographic — Living Robots Built Using Frog Cells